In this webinar, our speakers Dr. Carme Font, Prof. Tzu-Fei Wang, and Prof. Marc Carrier talked about the recent updates on cancer and thrombosis in the COVID-19 era.
They discussed:
• An update on the EPIPHANY study on serious complications prediction in patients with cancer
• Drug-drug interaction in the real-world setting of thrombosis and cancer
• Thromboprophylaxis dosing in COVID-19 patients
Prof. Ingrid Pabinger-Fasching chaired this event.
The recording and slides are available for our community members.
Register to our community to access them.
If you have missed our webinar or want to re-watch it, here are the event recording and our speakers’ slides. Enjoy!
ICTHIC Webinar recording:
Questions from the audience
Questions for Dr. Font:
Could you comment on DOAC vs LMWH treatment?
Almost all the patients included in the EPIPHANY received low-molecular-weight heparin (LMWH) during the first 3 months since the inclusions ended in 2014 when Direct Oral Anticoagulants (DOACs) were not yet used in this population. Some patients received fibrinolytic or unfractionated heparin.
Was there a risk stratification by an underlying cancer diagnosis (advanced, metastasis), other comorbidities in unsuspected PE?
We compared the baseline characteristics (Figure 1) according to the different proposed work-up scenarios (‘4S’ rule) in the prospectively assessed group of 497 patients.
Figure 1: baseline characteristics according to the different proposed work-up scenarios (‘4S’ rule) in the prospectively assessed group of 497 patients.
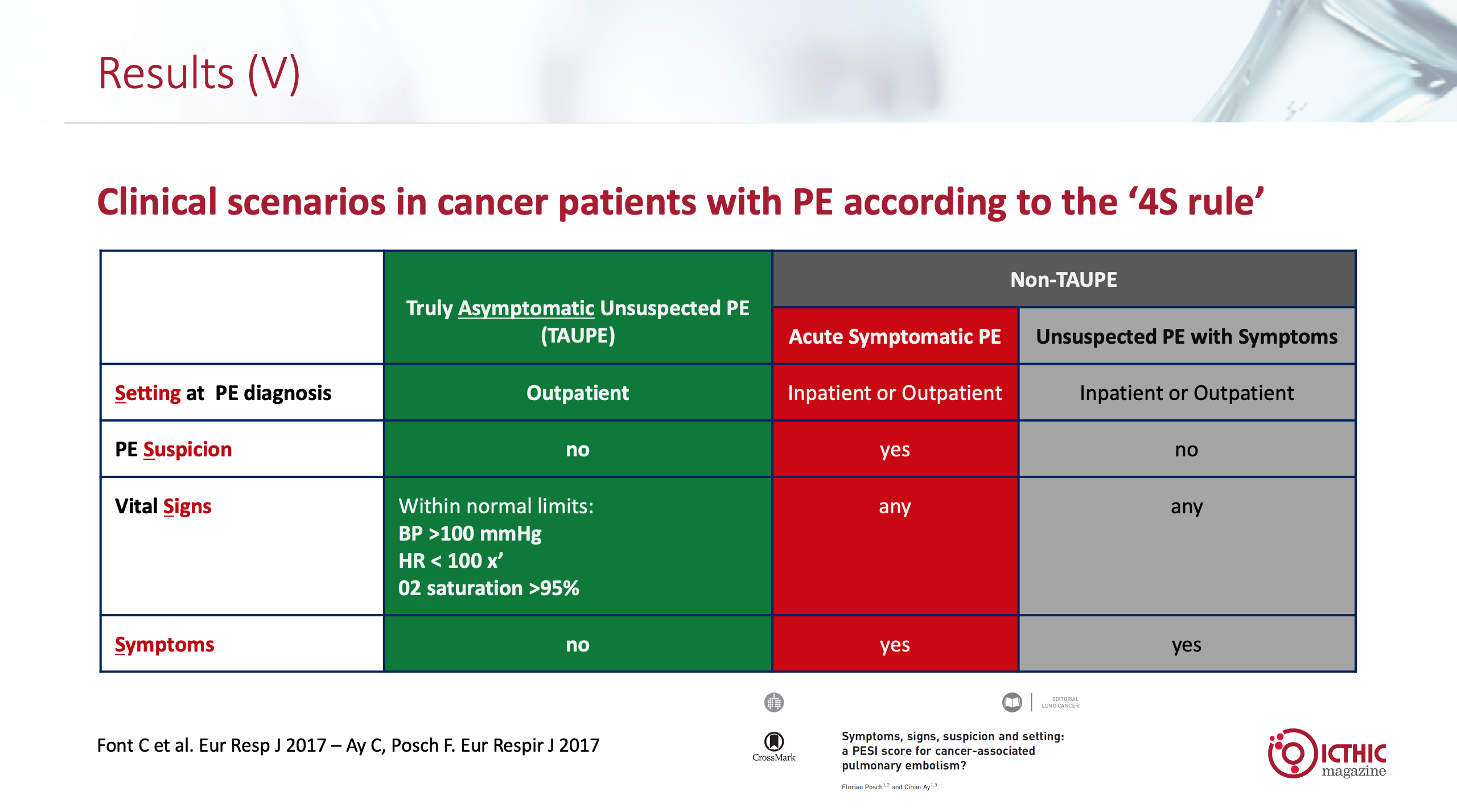
Of note, the proportion of metastatic disease was greater in patients with unsuspected PE (80% in Truly Asymptomatic Unsuspected Pulmonary Embolism, TA-UPE, and 86% in Unsuspected Pulmonary Embolism with Symptoms, UPE-S) compared to patients with symptomatic PE with 73% of patients with metastatic cancer (p=0.016; Figure 2).
Figure 2: Baseline characteristics in 497 prospectively-assessed patients according to work-up ascenarios

In the inclusion criteria for the study, we included patients with active cancer (localized or metastatic) or patients receiving adjuvant chemotherapy (no other adjuvant therapies or patients with cancer in remission).
Apart from the presence of symptoms, vital signs and inpatients vs outpatients, there were differences in the three groups regarding: Eastern Cooperative Oncology Group (ECOG) performance status (better in the TA-UPE group), surgery (greater in the symptomatic PE group), and chemotherapy and biological therapies, which were more common in the TA-UPE group.
Interestingly, no differences were observed regarding the comorbidities studied (chronic obstructive pulmonary disease, chronic heart disease, venous thromboembolism history).
We compared the 30-day mortality rates and the overall survival according to the three scenarios without risk stratification for other variables.
Was the location of the PE different between symptomatic vs asymptomatic? Did asymptomatic PE involve smaller branches of the pulmonary arteries?
In Figure 2, the radiological findings show that patients with acute symptomatic PE had bilateral lung involvement, multiple and troncular PE more frequently, whereas lobar and segmentary PE in more proximal locations were more frequent in the TA-UPE.
Question for Prof. Wang:
In terms of safety, should platelet count cut-off triggers for platelet transfusion be adjusted according to different drug–drug interactions and cancer/tumor location (i.e., brain, GI tract)?
This is certainly a great question and, this is commonly encountered in cancer patients. Unfortunately, we do not have strong evidence for the best management of anticoagulation in patients with thrombocytopenia, let alone when drug–drug interaction (DDI) is a concern. I can provide my personal practice but certainly, this is not the only answer, and am happy to hear what others have done. I typically follow the 2018 ISTH guidance document on the treatment of cancer-associated thrombosis in patients with thrombocytopenia (https://doi.org/10.1111/jth.14015), and without further data to support this, I have not routinely adjusted platelet cut-off triggers for specific tumor locations or DDIs. In patients where the concern of bleeding risks is high due to DDIs, typically change of potentially interacting drugs, if possible, could be considered, after discussion with patients and his/her oncologist.
Questions for Prof. Carrier:
Is there any advocacy for half therapeutic dosing (75–100 IU/kg once daily) in COVID-19 ICU patients?
No randomized controlled trial evidence to support half therapeutic dosing in COVID-19 ICU patients, but it is controversial. The REMAP-CAP group is currently carrying out a randomized trial comparing prophylactic to intermediate dosing in COVID-19 ICU patients. Hopefully, we will have prospective data soon.
What is your opinion regarding the use of DOACs in these patients?
I would only use DOACs if the patients are already on a DOAC at the time of hospitalization for COVID (e.g., atrial fibrillation). If the patient is not already on anticoagulation, I use LMWH (or unfractionated heparin) for thromboprophylaxis. Heparinoids may have additional benefits than just anticoagulation (decrease viral entry, NETs, etc).
These results are largely unexpected. Based on virtually all uncontrolled studies so far available, most international guidelines recommend against full-dose anticoagulation in COVID-19 patients free from thrombosis. Do you think that these studies have the potential to change clinical practice?
Yes. I think that upon publication, clinical practice guidelines will change. The pre-print manuscript should be available in a couple of weeks.
How do you adjust the heparin dose in the presence of thrombocytopenia?
I do not use therapeutic dosing of LMWH in patients with platelet counts <50. Intermediate or prophylactic dosing is reasonable in patients with platelet counts between 20 and 50. Anticoagulation should be held in platelet counts <20.
What do you think about aspirin in the pre-hospitalization phase?
It is a good hypothesis. Many studies are currently assessing anti-platelets in different settings (ICU, moderately ill, pre- and post-hospitalization). Hopefully, we will have more data soon.















